Oct. 29, 2018
VCU Health investigates stem-cell-based therapy to treat heart failure
Share this story
Heart failure, a condition commonly treated with lifelong medication, affects 5.7 million Americans. The American Heart Association projects that by 2030, those suffering from the condition will rise to 8 million. The VCU Health Pauley Heart Center is participating in a new clinical trial to explore the body’s natural healing power through stem cells, which could one day reduce or eliminate the need for medication to treat ischemic heart failure after a heart attack.
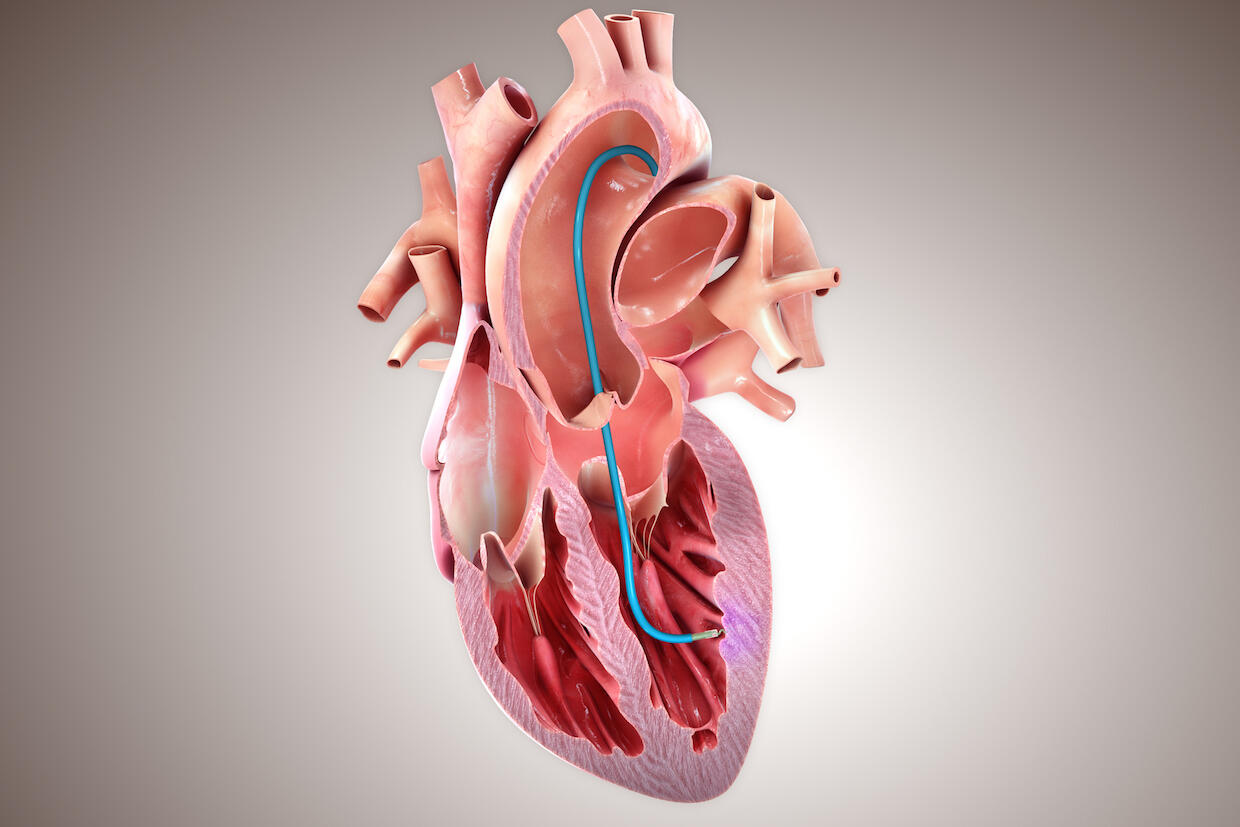
The CardiAMP Heart Failure clinical trial of an investigational stem-cell-based therapy takes a personalized and minimally invasive approach by using a patient’s own cells in the treatment.
“As part of an academic medical center, we are constantly exploring innovative procedures for our patients,” said William Gregory Hundley, M.D., director of the Pauley Heart Center. “We have just enrolled our first two patients into the trial. By participating in research for a condition that affects so many, we have the potential to really improve the quality of life for a large population of patients.”
About 790,000 people in the U.S. have heart attacks each year, according to the American Heart Association.
“We’re looking at how use of a patient’s own stem cells promotes self-healing within his or her heart,” said Zachary Gertz, M.D., interventional cardiologist at the Pauley Heart Center. “Oftentimes, advanced heart failure patients need an artificial pump or a heart transplant. With this trial, we hope to identify another option to avoid such an invasive step.
Patients are screened for bone marrow characteristics that could result in a higher likelihood of them benefiting from the therapy. Likely responders who are randomized into the treatment arm of the trial receive the therapy, which includes a high dose of mononuclear cells directly to damaged regions of the heart.
Clinical studies of CardiAMP investigational therapy to date have shown clinically meaningful and sustained improvements in quality of life and functional capacity for patients with few risks. The CardiAMP Heart Failure trial is a Phase III study of up to 260 patients at up to 40 centers nationwide.
The Pauley Heart Center continues to enroll patients in the trial. For information about eligibility or enrollment, please visit studyfinder.cctr.vcu.edu or Clinicaltrials.gov.
Subscribe to VCU News
Subscribe to VCU News at newsletter.vcu.edu and receive a selection of stories, videos, photos, news clips and event listings in your inbox.