
May 14, 2026
VCU innovation may prevent scar tissue and advance joint surgery
Share this story
As patients recover from joint surgery or traumatic injury, the body’s repair process might go into overdrive, producing excessive scar tissue inside the joint. The result – arthrofibrosis – can lead to pain, stiffness and a loss of mobility that is difficult to reverse once it begins.
Despite how common and debilitating arthrofibrosis can be, there are currently no reliable ways to predict who will develop it, nor any effective treatments once it takes hold.
“Once it’s underway, it’s pretty much underway,” said Barbara D. Boyan, Ph.D., executive director of Virginia Commonwealth University’s Institute for Engineering and Medicine and the Alice T. and William H. Goodwin Jr. Professor in the College of Engineering’s Department of Biomedical Engineering.
Boyan and her team at VCU’s Laboratory for Musculoskeletal Research and Innovation are taking a novel approach. Rather than attempting to treat arthrofibrosis after scar tissue has formed, their innovation focuses on preventing it from developing in the first place, intervening at the moment when inflammation begins – during joint surgery itself.
If successful, the impact could be felt far beyond individual patients – reshaping standards of care, reducing the need for follow-up surgeries and improving quality of life for countless people recovering from joint procedures.
A breakthrough in gel form
The work builds on years of foundational research conducted at LMRI, where Boyan’s group studies disorders of the musculoskeletal system, including bones, cartilage and joints.
Long before their current intervention took shape, the lab had identified unresolved inflammation as a key driver of fibrotic scar formation. The team was also intrigued by evidence showing that arthrofibrosis occurs more frequently and more severely in women, suggesting underlying biological differences in immune response.
A breakthrough came with the development of ClickGel, an injectable biomaterial originally designed for neurosurgical applications by Pascal Medical Corp., which Boyan co-founded. ClickGel is delivered as a liquid and then rapidly forms a stable matrix in the body, allowing it to remain precisely where it is placed without interfering with surrounding tissue.
Pascal won an $800,000 state grant in 2025 to support ClickGel’s development, while VCU TechTransfer and Ventures helped Boyan’s team protect their intellectual property and connected them to business advisors.
“We looked at ClickGel’s properties and how it’s very easy to inject compared to other carrier systems,” Boyan said.
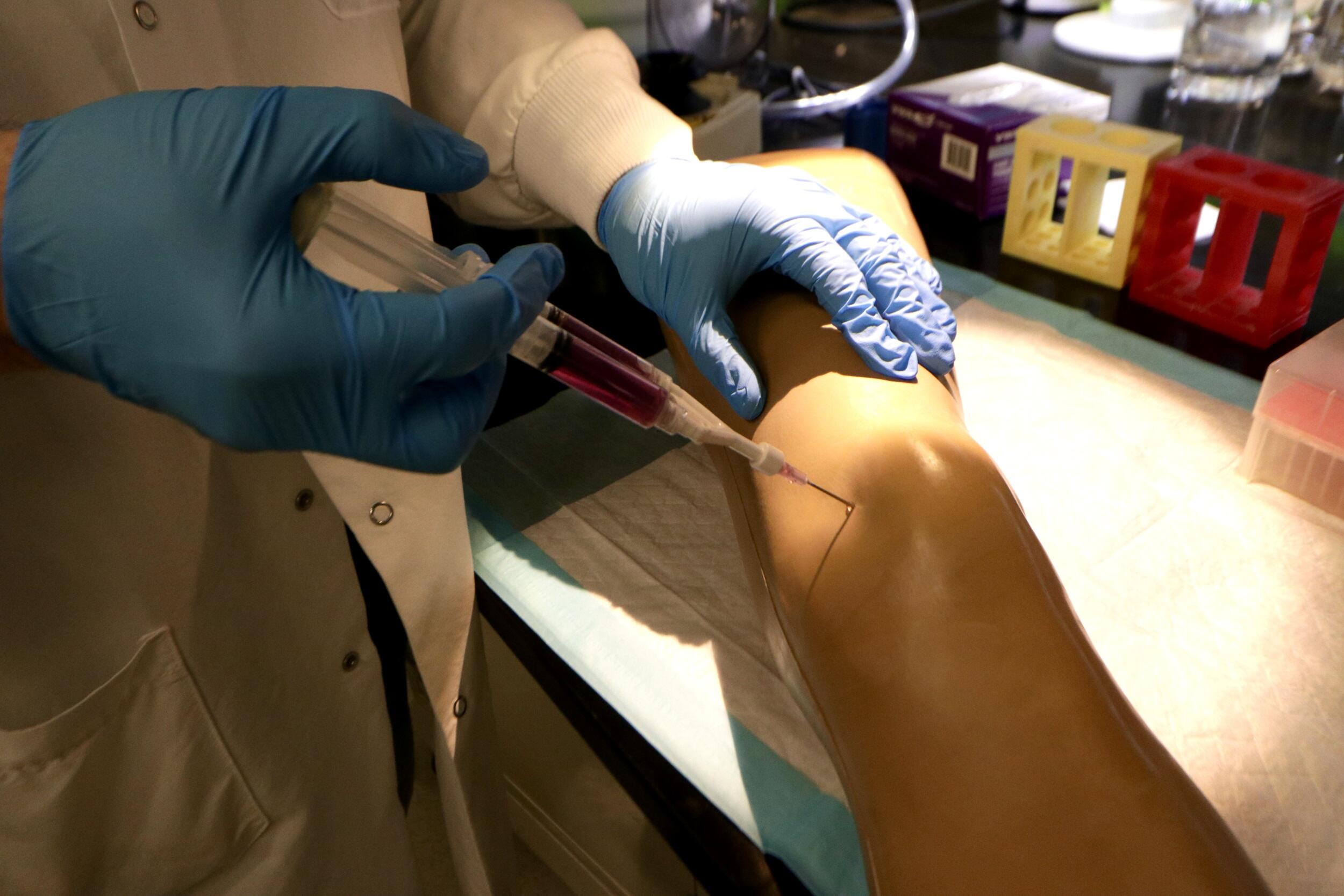
The ideal time and place for treatment
She and her team then had the idea to place an anti-inflammatory therapeutic in the gel that would prevent arthrofibrosis, then inject the substance into the joint of a surgery patient before anesthesia wears off. By resolving inflammation early, the intervention aims to stop the cascade that leads to excessive scar formation.
Previous attempts to address arthrofibrosis have fallen short for various reasons. Anti-inflammatory drugs injected on their own disperse too quickly to be effective. Other carrier materials have struggled with the tight physical constraints of joint spaces.
“Whatever you put in the joint has to be a liquid when it goes in, and it has to then firm up without interfering with articulating joints,” Boyan said.
Her approach overcomes these challenges by combining an injectable gel with sustained local delivery of lipid nanoparticles to deliver microRNAs that regulate inflammation and collagen production.
Progress toward commercialization
The project recently received support from the TechTransfer and Ventures Commercialization Fund, which will help advance the technology toward clinical use. ClickGel has already been designated by the Food and Drug Administration as a medical device and is on its way to commercialization as a dural sealant at Pascal.
“The first thing we have to do is get approval to use ClickGel in humans as a device without any additions added,” Boyan said.
Once approved, her team can pursue a combination product that incorporates anti-inflammatory therapeutics for joint applications, a process she estimates could take about five years.
The commercialization effort is supported by ongoing collaboration with TechTransfer and Ventures, part of VCU’s Office of the Vice President for Research and Innovation. Director of Licensing Magdalena K. Morgan, Ph.D., works closely with Boyan’s team to align scientific development with regulatory and market realities.
“Dr. Boyan’s approach is a perfect example of strategic innovation,” Morgan said. “By taking a device that is already moving through the FDA pipeline and adapting it for therapeutic delivery, the team is significantly shortening the path to getting a solution into the hands of surgeons.”
Team bridges research and medicine
Working directly on the arthrofibrosis intervention project with Boyan are D. Joshua Cohen, M.D., research assistant professor in the Department of Biomedical Engineering, and LMRI doctoral student Kait Hosmer.
As a physician-scientist, Cohen – who also serves as an animal models consultant with Pascal – combines clinical perspective with deep musculoskeletal and biomaterials knowledge to inform real-world surgical integration. He is a co-inventor of the technology and has developed the model systems the team is using to design and validate its use to prevent arthrofibrosis. Hosmer, meanwhile, contributes to experimental design and translational research.
Boyan and her collaborators envision a future in which arthrofibrosis is no longer an accepted risk of joint surgery but a complication that can be proactively avoided.
“Pascal’s hydrogel is an exceptional platform for delivering therapeutics that need to work locally and over sustained periods of time,” Boyan said. “If we find that it enables us to develop a treatment that prevents or reduces fibrosis in joints, we will be able to tackle a lot of problems plaguing musculoskeletal health.”
Subscribe to VCU News
Subscribe to VCU News at newsletter.vcu.edu and receive a selection of stories, videos, photos, news clips and event listings in your inbox.










