Dec. 12, 2018
Clearest view ever of cell membrane yields unexpected structure, new research possibilities
Share this story
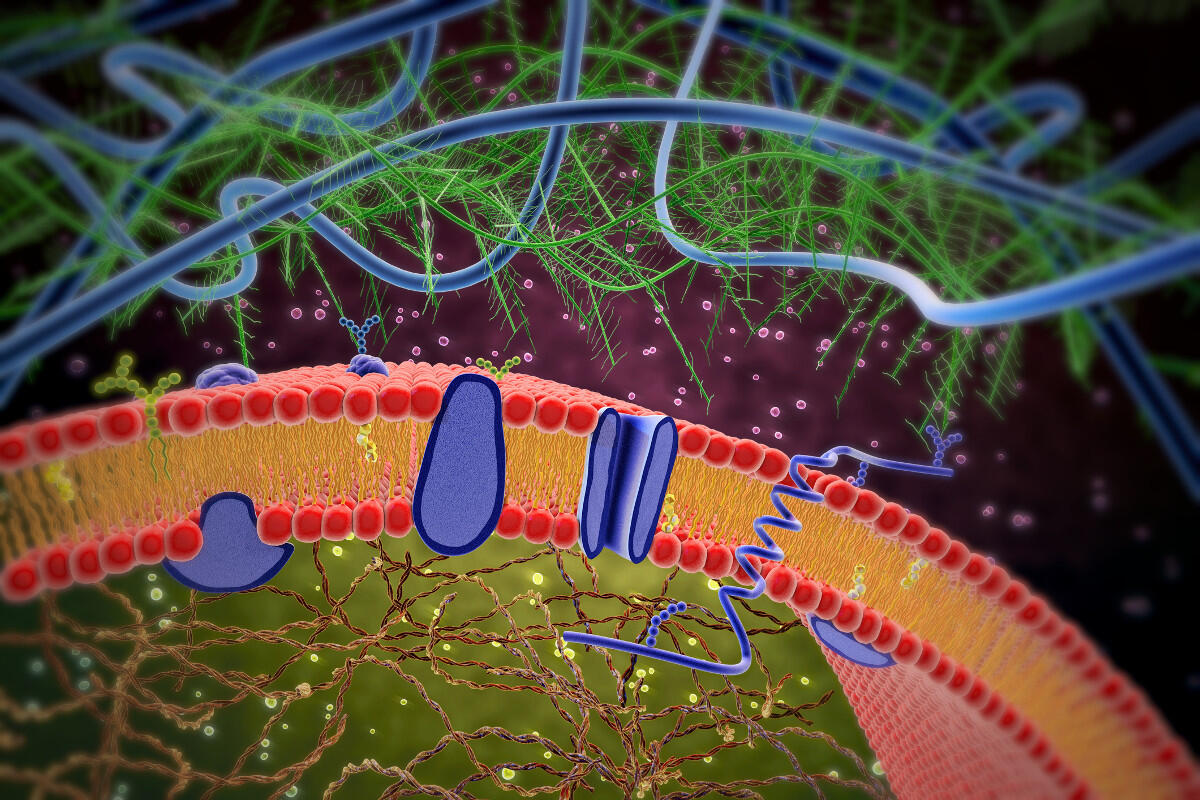
Working with a Nobel Prize-winning biophysicist, a team of researchers at Virginia Commonwealth University has gained the clearest view yet of a patch of cell membrane and its components, revealing unexpected structures and opening up new possibilities for pharmaceutical research.
Cell membranes are formed largely of a bimolecular sheet, a fraction of the thickness of a soap bubble, in which two layers of lipid molecules are packed with their hydrophobic tails pointing inward and their hydrophilic heads outward, exposed to water.

The internal shape and structure of this lipid bilayer have remained largely mysterious after almost a century of research. This is in large part because most methods to examine membranes use detergents, which strip away the lipids that make up much of the membranes’ structures.
In a newly published paper in Proceedings of the National Academy of Sciences of the United States of America, the team — led by Youzhong Guo, Ph.D., of VCU's School of Pharmacy — used a new detergent-free method that allowed them to examine the membrane of an E. coli cell, with lipids still in place.
Where earlier models had shown a fluid, almost structureless lipid layer — one often-cited research paper compared it to different weights of olive oil poured together — the VCU-led team was startled to find a distinct hexagonal structure inside the membrane. This led the researchers to propose that the lipid layer might act as both sensor and energy transducer within a membrane-protein transporter.
“The most surprising outcome is the high order with which lipid molecules are arranged, and the idea they might even cooperate in the functional cycle of the export channel,” said Joachim Frank, Ph.D., of Columbia University, a 2017 Nobel laureate in chemistry and co-author of the paper. “It is counterintuitive since we have learned that lipids are fluid and disordered in the membrane.”
The researchers were able to get such a clear view because they used an innovative method to isolate and stabilize the membranes. Employing poly-styrene-maleic acid to break cell membranes into nanoparticles that were then isolated and captured in a layer of sophisticated polymer, the researchers used the cryo-electron microscope at the New York Structural Biology Center to get a clear look at the lipid bilayer.
“Being able to pull proteins out of cell membranes without using detergents to break up the lipid bilayers truly is a fantastic advance,” said Wayne Hendrickson, Ph.D., university professor at Columbia, scientific director of the New York Structural Biology Center and co-author of the paper.
The technique and its revelations could have significant pharmaceutical value, Guo said. He said about half of medical drugs target the cell membranes, and proposed that improved understanding of their layers of lipids and proteins could lead to new or more effective therapies.
The authors of the paper, “Structure and Activity of Lipid Bilayer within a Membrane-Protein Transporter,” are VCU’s Guo, Weihua Qiu, Ph.D.; Guoyan G. Xu, Ph.D.; and Yan Zhang, Ph.D.; and Columbia’s Frank, Hendrickson, Ziao Fu, Ph.D.; and Robert A. Grassucci, Ph.D.
Subscribe to VCU News
Subscribe to VCU News at newsletter.vcu.edu and receive a selection of stories, videos, photos, news clips and event listings in your inbox.










